Color and Palette Design with VISTA
VISTA Development Team
VISTA-colors.RmdOverview
A common challenge in RNA-seq analysis is visual consistency: the same biological group should keep the same color across PCA, MDS, expression, and summary plots. When this is not controlled, interpretation becomes harder and figure legends can become confusing.
This vignette focuses on VISTA’s color system and shows how to:
- Discover all built-in palette options.
- Keep colors consistent across different plot families.
- Change global color style in one place.
- Apply manual group/comparison color maps when required.
The floating table of contents on the side provides a quick-scroll panel for navigating sections.
Load Packages and Data
library(VISTA)
library(ggplot2)
library(dplyr)
library(tibble)
library(colorspace)
data("count_data", package = "VISTA")
data("sample_metadata", package = "VISTA")
# Keep vignette runtime moderate while preserving signal
count_small <- count_data[1:1500, ]
# Use all available samples
table(sample_metadata$cond_long)
#>
#> control treatment1
#> 4 4Build a Baseline VISTA Object
vista_dark <- create_vista(
counts = count_small,
sample_info = sample_metadata,
column_geneid = "gene_id",
group_column = "cond_long",
group_numerator = "treatment1",
group_denominator = "control",
group_palette = "Dark 2",
comparison_palette = "Dark 3",
min_counts = 5,
min_replicates = 1
)
vista_dark
#> class: VISTA
#> dim: 1305 8
#> metadata(12): de_results de_summary ... design comparison
#> assays(1): norm_counts
#> rownames(1305): ENSG00000000003 ENSG00000000419 ... ENSG00000080007
#> ENSG00000080031
#> rowData names(1): baseMean
#> colnames(8): SRR1039508 SRR1039509 ... SRR1039520 SRR1039521
#> colData names(14): SampleName cell ... sizeFactor sample_namesInspect the stored palette metadata:
group_palette(vista_dark)
#> [1] "Dark 2"
group_colors(vista_dark)
#> control treatment1
#> "#C87A8A" "#00A396"Built-In Palette Choices
VISTA group palettes are based on colorspace qualitative HCL palettes.
palette_table <- colorspace::hcl_palettes(type = "qualitative") |>
as.data.frame() |>
tibble::rownames_to_column("palette") |>
dplyr::select(palette)
n_palettes <- nrow(palette_table)
cat("Number of built-in qualitative palettes available in VISTA:", n_palettes, "\n\n")
#> Number of built-in qualitative palettes available in VISTA: 9
palette_table
#> palette
#> 1 Pastel 1
#> 2 Dark 2
#> 3 Dark 3
#> 4 Set 2
#> 5 Set 3
#> 6 Warm
#> 7 Cold
#> 8 Harmonic
#> 9 DynamicCreate a visual palette atlas:
palette_grid <- do.call(
rbind,
lapply(palette_table$palette, function(pal) {
cols <- colorspace::qualitative_hcl(8, palette = pal)
data.frame(
palette = pal,
idx = seq_along(cols),
col = cols,
stringsAsFactors = FALSE
)
})
)
palette_grid$palette <- factor(palette_grid$palette, levels = rev(palette_table$palette))
ggplot(palette_grid, aes(x = idx, y = palette, fill = col)) +
geom_tile(color = "white", linewidth = 0.3) +
scale_fill_identity() +
scale_x_continuous(breaks = 1:8) +
labs(
x = "Color index within palette",
y = NULL,
title = "VISTA Palette Atlas (Qualitative HCL)"
) +
theme_minimal(base_size = 12) +
theme(panel.grid = element_blank())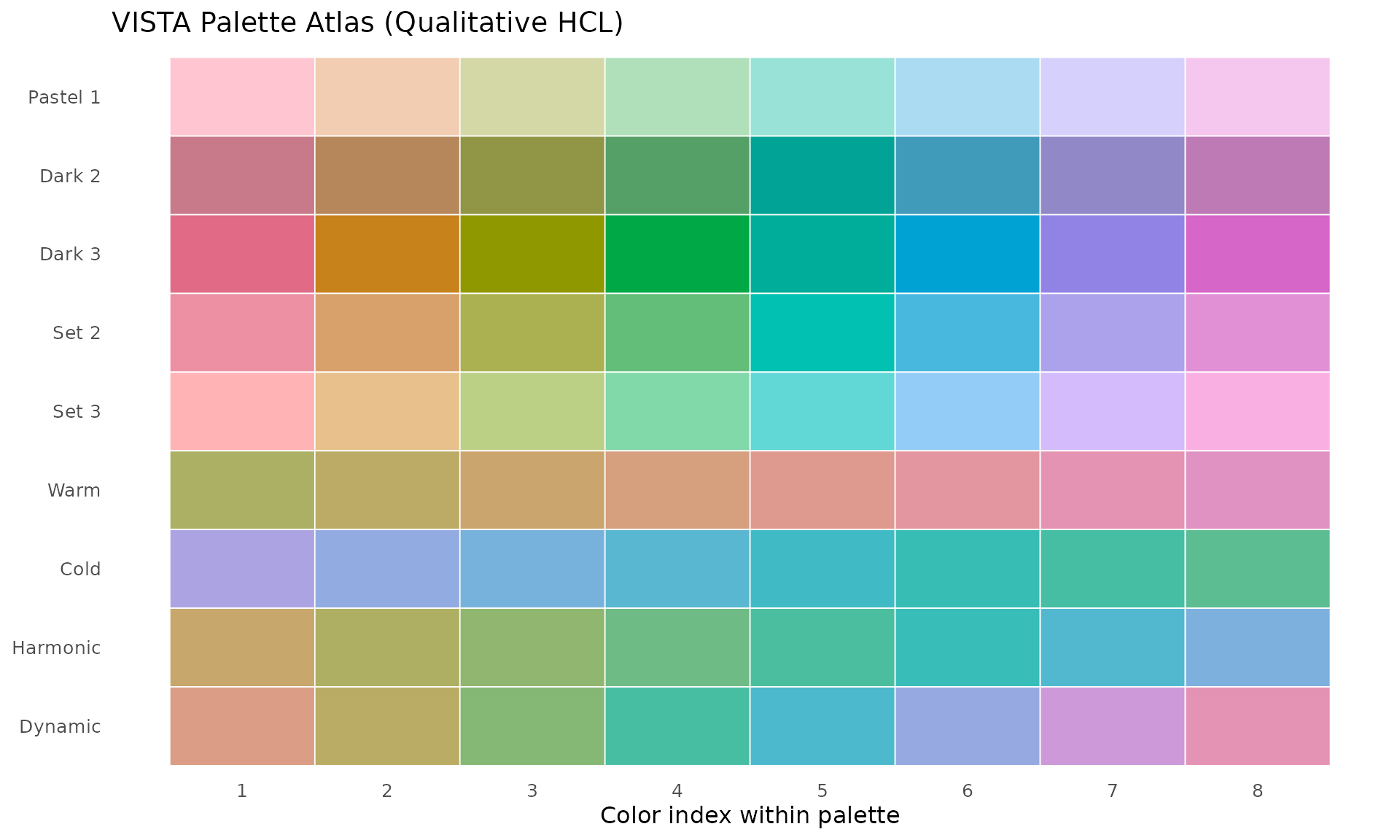
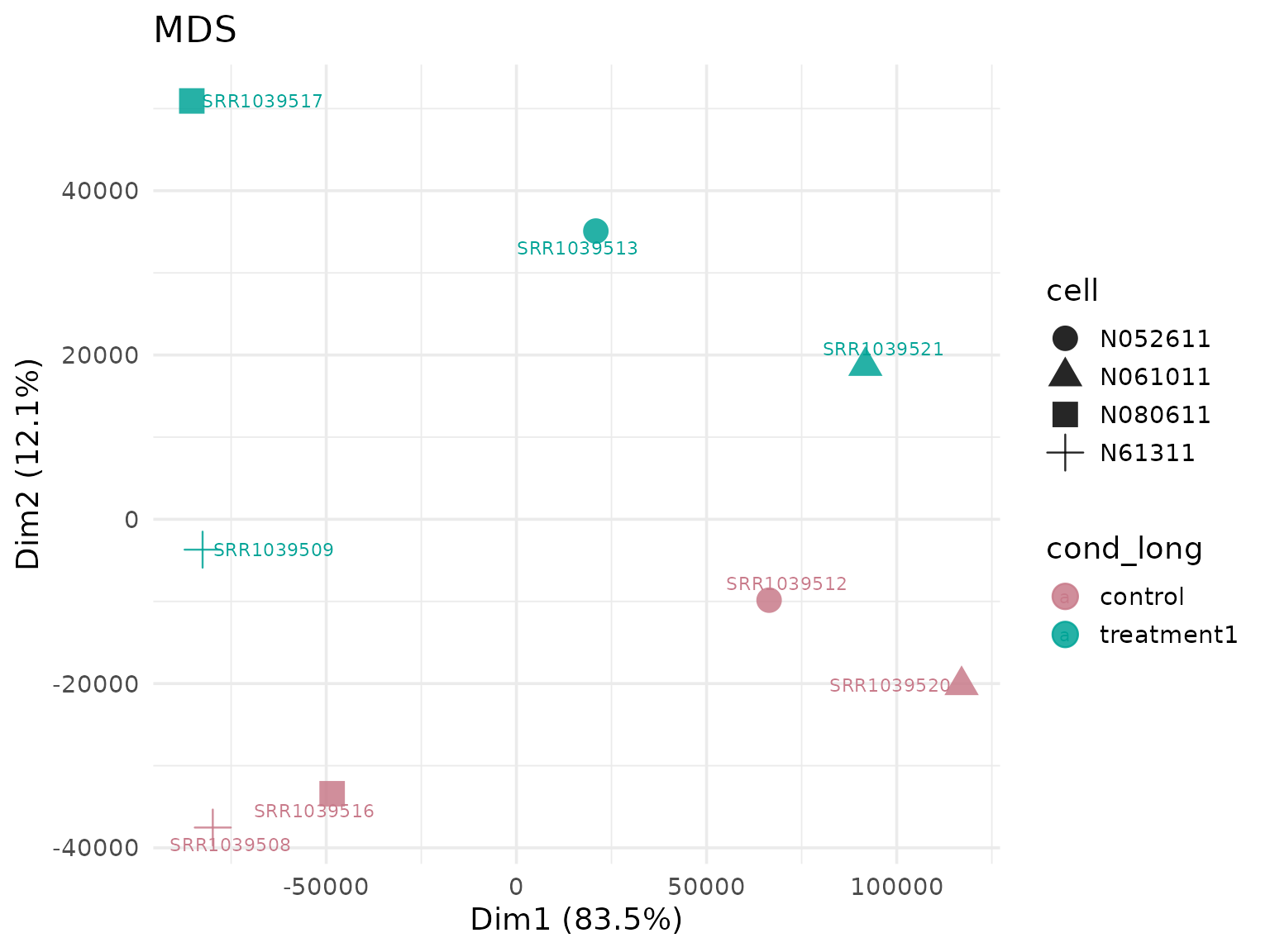
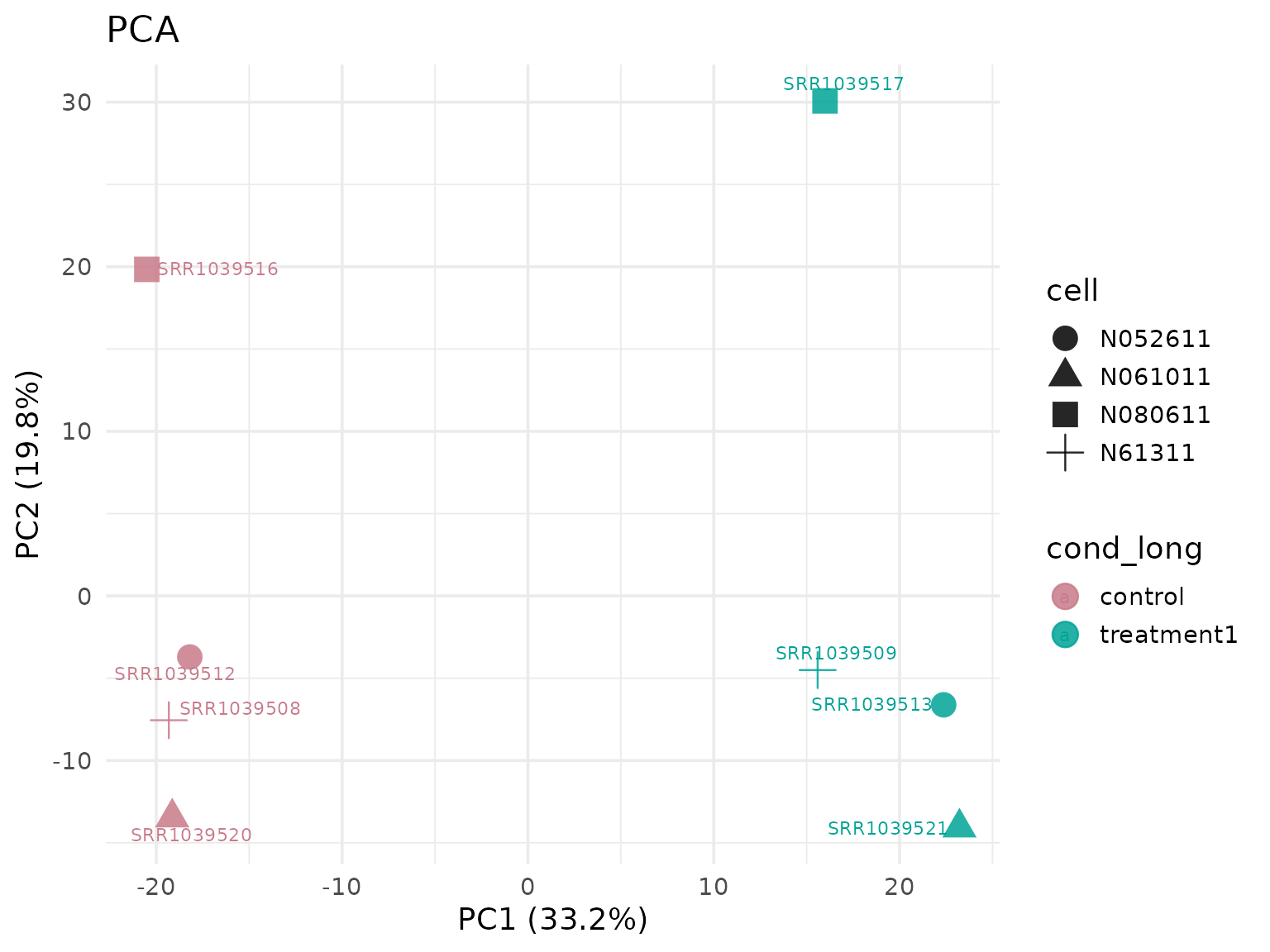
Consistency Across Plot Families
Use the same VISTA object and verify that group colors
are preserved across multiple visualization types.
comp_name <- names(comparisons(vista_dark))[1]
up_genes <- get_genes_by_regulation(
vista_dark,
sample_comparisons = comp_name,
regulation = "Up"
)[[comp_name]]
if (length(up_genes) < 6) {
genes_demo <- head(rownames(vista_dark), 6)
} else {
genes_demo <- head(up_genes, 6)
}
genes_demo
#> [1] "ENSG00000003402" "ENSG00000004799" "ENSG00000006788" "ENSG00000008256"
#> [5] "ENSG00000008311" "ENSG00000009413"PCA (group colors from object metadata)
get_pca_plot(
vista_dark,
label = TRUE,
shape_by = "cell",
point_size = 5
)
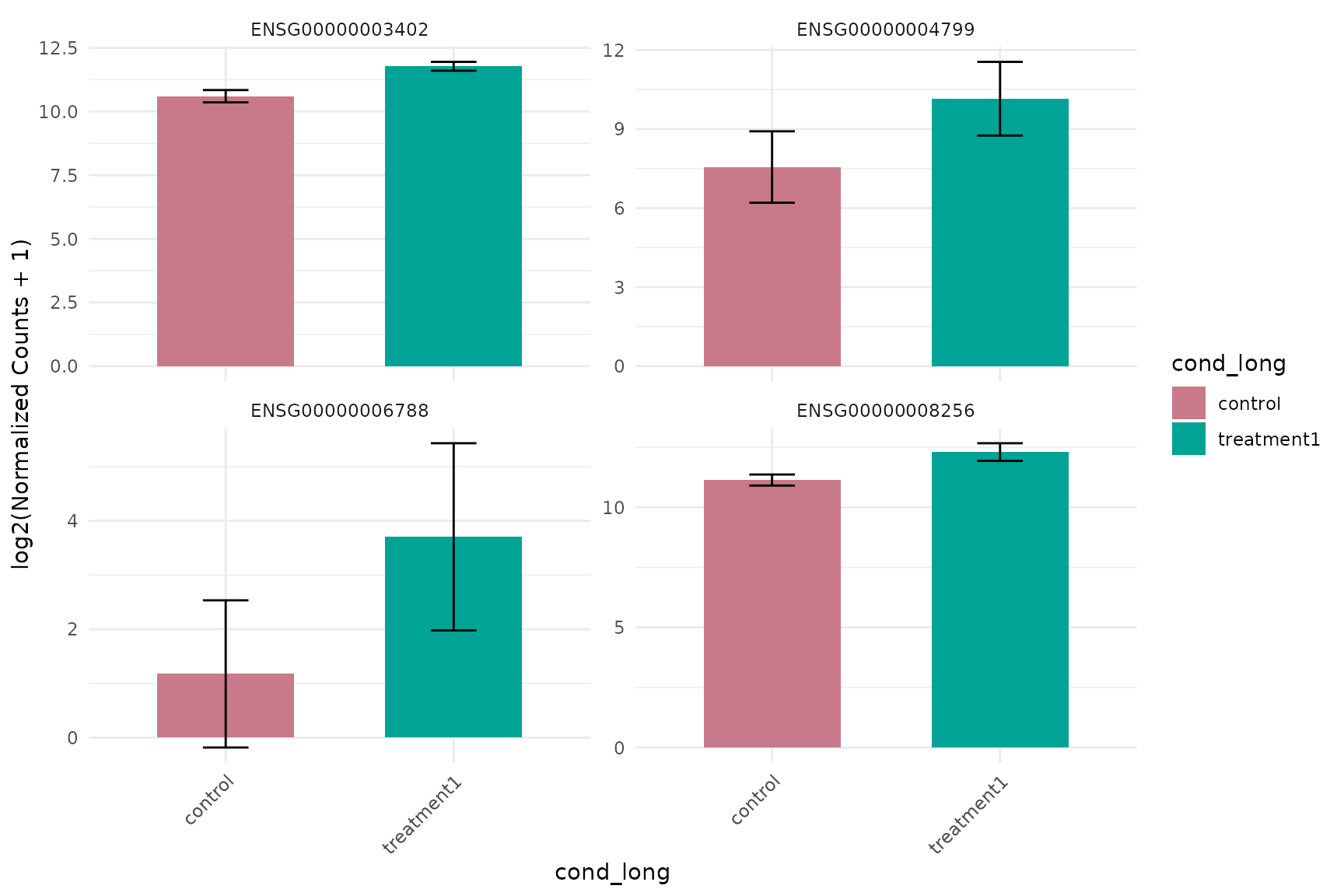
Expression barplot (same group colors)
get_expression_barplot(
vista_dark,
genes = genes_demo[1:4],
log_transform = TRUE
)
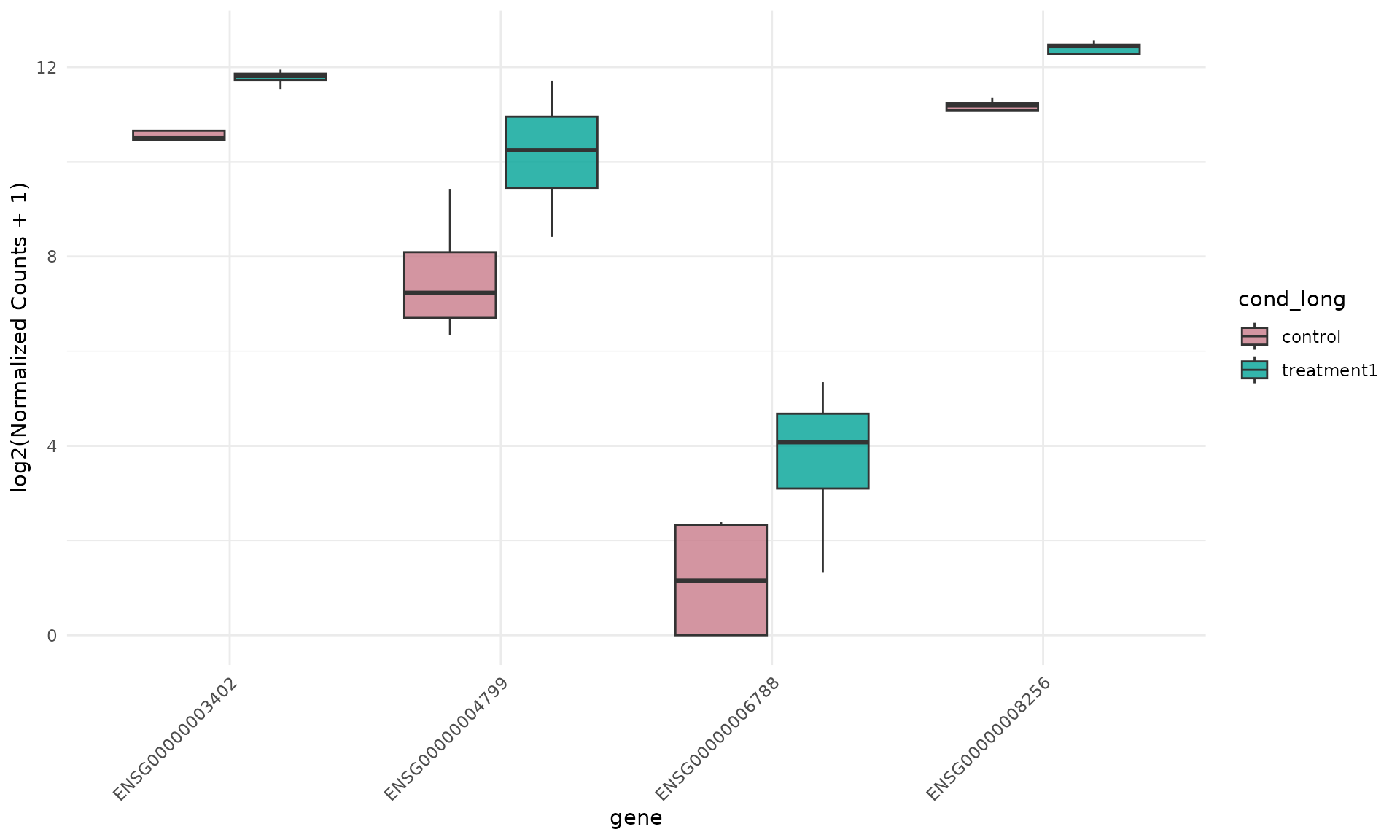
Expression boxplot (same group colors)
get_expression_boxplot(
vista_dark,
genes = genes_demo[1:4],
log_transform = TRUE,
pool_genes = FALSE,
by = "gene",
facet_by = "none",
fill_by = "group"
)
Switching Global Style in One Argument
Rebuild with a different global palette. No plot-specific color edits are needed.
vista_warm <- create_vista(
counts = count_small,
sample_info = sample_metadata,
column_geneid = "gene_id",
group_column = "cond_long",
group_numerator = "treatment1",
group_denominator = "control",
group_palette = "Warm",
comparison_palette = "Set 3",
min_counts = 5,
min_replicates = 1
)
group_palette(vista_warm)
#> [1] "Warm"
group_colors(vista_warm)
#> control treatment1
#> "#ABB065" "#E093C3"
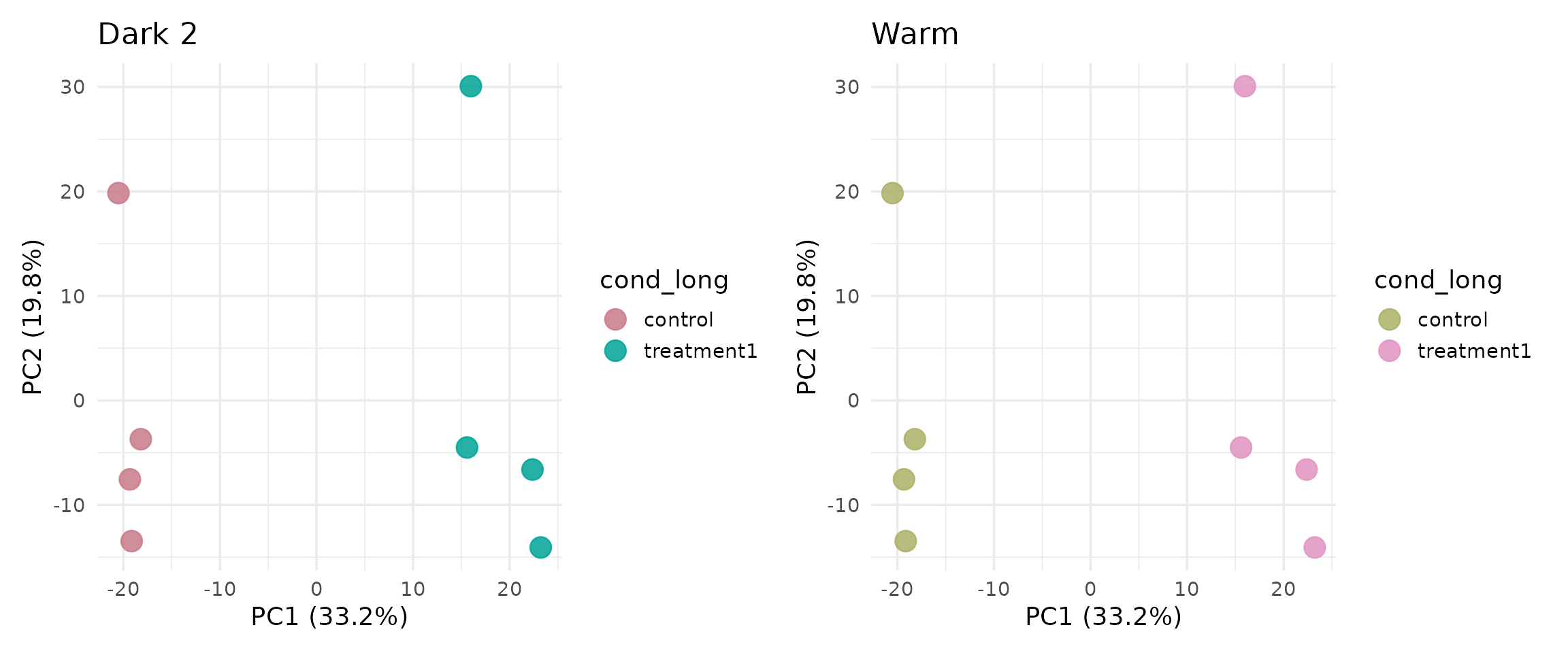
p_dark <- get_pca_plot(vista_dark, label = FALSE, point_size = 5) +
ggtitle("Dark 2")
p_warm <- get_pca_plot(vista_warm, label = FALSE, point_size = 5) +
ggtitle("Warm")
if (requireNamespace("patchwork", quietly = TRUE)) {
patchwork::wrap_plots(p_dark, p_warm, ncol = 2)
} else {
p_dark
p_warm
}
Comparison-Level Colors
For multi-comparison workflows, VISTA also stores comparison colors.
# Build two directed comparisons to demonstrate comparison color mapping
vista_multi <- create_vista(
counts = count_small,
sample_info = sample_metadata,
column_geneid = "gene_id",
group_column = "cond_long",
group_numerator = c("treatment1", "control"),
group_denominator = c("control", "treatment1"),
group_palette = "Dark 2",
comparison_palette = "Set 3",
min_counts = 5,
min_replicates = 1
)
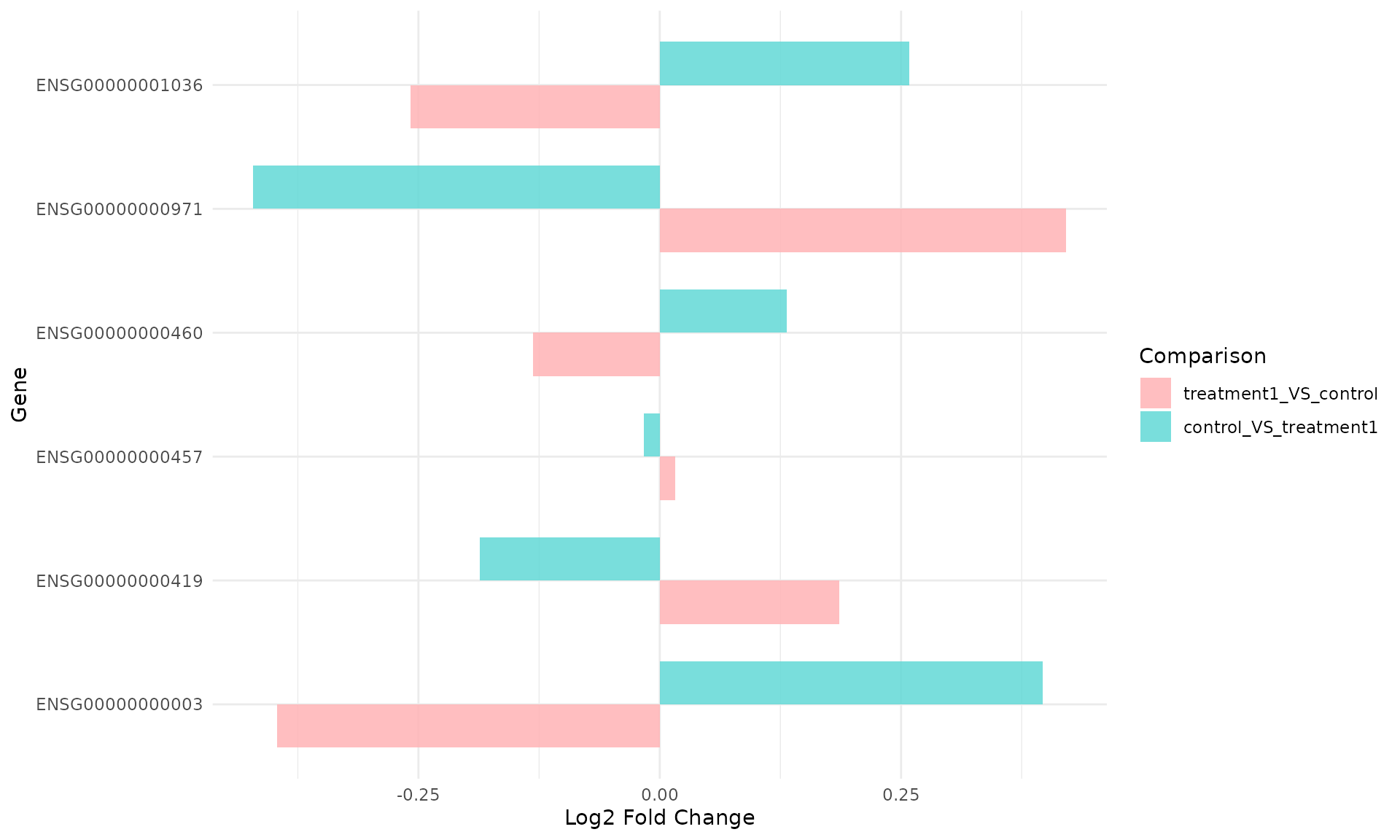
S4Vectors::metadata(vista_multi)$comparison$colors
#> treatment1_VS_control control_VS_treatment1
#> "#FFB3B5" "#61D8D6"
get_foldchange_barplot(
vista_multi,
genes = head(rownames(vista_multi), 6),
facet_by = "none"
)
Manual Color Maps (Advanced)
If your lab or journal has fixed brand colors, you can set explicit named vectors using built-in VISTA setters.
Available manual setters:
set_vista_group_colors(vista, color_map)set_vista_comparison_colors(vista, color_map)
custom_group_cols <- c(
control = "#264653",
treatment1 = "#E76F51"
)
custom_comp_cols <- c(
treatment1_VS_control = "#6C5CE7",
control_VS_treatment1 = "#00A896"
)
vista_custom <- set_vista_group_colors(vista_dark, custom_group_cols)
vista_multi_custom <- set_vista_comparison_colors(vista_multi, custom_comp_cols)
group_colors(vista_custom)
#> control treatment1
#> "#264653" "#E76F51"
S4Vectors::metadata(vista_multi_custom)$comparison$colors
#> treatment1_VS_control control_VS_treatment1
#> "#6C5CE7" "#00A896"
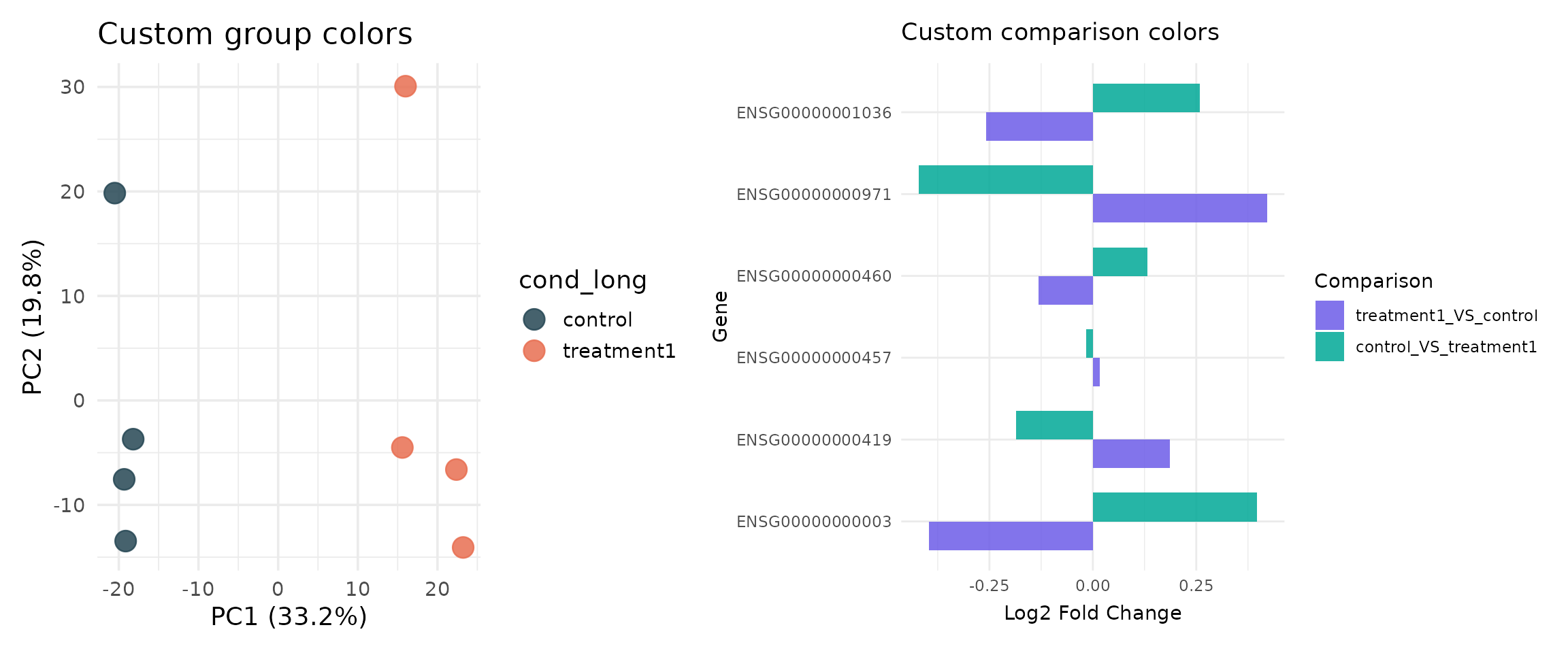
p_custom_group <- get_pca_plot(
vista_custom,
label = FALSE,
point_size = 5
) + ggtitle("Custom group colors")
p_custom_comp <- get_foldchange_barplot(
vista_multi_custom,
genes = head(rownames(vista_multi_custom), 6),
facet_by = "none"
) + ggtitle("Custom comparison colors")
if (requireNamespace("patchwork", quietly = TRUE)) {
patchwork::wrap_plots(p_custom_group, p_custom_comp, ncol = 2)
} else {
p_custom_group
p_custom_comp
}
Practical Guidance
- Use
group_paletteandcomparison_paletteincreate_vista()for fast, global styling. - Use
group_colors(vista)to audit mapping before generating publication panels. - Keep one palette per manuscript/project to preserve interpretation consistency.
- Switch to manual named vectors only when you need strict journal or brand colors.
Session Information
sessionInfo()
#> R version 4.5.3 (2026-03-11)
#> Platform: x86_64-pc-linux-gnu
#> Running under: Ubuntu 24.04.4 LTS
#>
#> Matrix products: default
#> BLAS: /usr/lib/x86_64-linux-gnu/openblas-pthread/libblas.so.3
#> LAPACK: /usr/lib/x86_64-linux-gnu/openblas-pthread/libopenblasp-r0.3.26.so; LAPACK version 3.12.0
#>
#> locale:
#> [1] LC_CTYPE=C.UTF-8 LC_NUMERIC=C LC_TIME=C.UTF-8
#> [4] LC_COLLATE=C.UTF-8 LC_MONETARY=C.UTF-8 LC_MESSAGES=C.UTF-8
#> [7] LC_PAPER=C.UTF-8 LC_NAME=C LC_ADDRESS=C
#> [10] LC_TELEPHONE=C LC_MEASUREMENT=C.UTF-8 LC_IDENTIFICATION=C
#>
#> time zone: UTC
#> tzcode source: system (glibc)
#>
#> attached base packages:
#> [1] stats graphics grDevices utils datasets methods base
#>
#> other attached packages:
#> [1] colorspace_2.1-2 tibble_3.3.1 dplyr_1.2.0 ggplot2_4.0.2
#> [5] VISTA_0.99.4 BiocStyle_2.38.0
#>
#> loaded via a namespace (and not attached):
#> [1] RColorBrewer_1.1-3 jsonlite_2.0.0
#> [3] tidydr_0.0.6 magrittr_2.0.4
#> [5] ggtangle_0.1.1 farver_2.1.2
#> [7] rmarkdown_2.31 fs_2.0.1
#> [9] ragg_1.5.2 vctrs_0.7.2
#> [11] memoise_2.0.1 ggtree_4.0.5
#> [13] rstatix_0.7.3 htmltools_0.5.9
#> [15] S4Arrays_1.10.1 curl_7.0.0
#> [17] broom_1.0.12 Formula_1.2-5
#> [19] SparseArray_1.10.10 gridGraphics_0.5-1
#> [21] sass_0.4.10 bslib_0.10.0
#> [23] htmlwidgets_1.6.4 desc_1.4.3
#> [25] plyr_1.8.9 cachem_1.1.0
#> [27] igraph_2.2.2 lifecycle_1.0.5
#> [29] pkgconfig_2.0.3 gson_0.1.0
#> [31] Matrix_1.7-4 R6_2.6.1
#> [33] fastmap_1.2.0 MatrixGenerics_1.22.0
#> [35] digest_0.6.39 aplot_0.2.9
#> [37] enrichplot_1.30.5 ggnewscale_0.5.2
#> [39] GGally_2.4.0 patchwork_1.3.2
#> [41] AnnotationDbi_1.72.0 S4Vectors_0.48.0
#> [43] DESeq2_1.50.2 textshaping_1.0.5
#> [45] GenomicRanges_1.62.1 RSQLite_2.4.6
#> [47] ggpubr_0.6.3 labeling_0.4.3
#> [49] polyclip_1.10-7 httr_1.4.8
#> [51] abind_1.4-8 compiler_4.5.3
#> [53] withr_3.0.2 bit64_4.6.0-1
#> [55] fontquiver_0.2.1 backports_1.5.0
#> [57] S7_0.2.1 BiocParallel_1.44.0
#> [59] carData_3.0-6 DBI_1.3.0
#> [61] ggstats_0.13.0 ggforce_0.5.0
#> [63] R.utils_2.13.0 ggsignif_0.6.4
#> [65] MASS_7.3-65 rappdirs_0.3.4
#> [67] DelayedArray_0.36.0 tools_4.5.3
#> [69] otel_0.2.0 scatterpie_0.2.6
#> [71] ape_5.8-1 msigdbr_26.1.0
#> [73] R.oo_1.27.1 glue_1.8.0
#> [75] nlme_3.1-168 GOSemSim_2.36.0
#> [77] grid_4.5.3 cluster_2.1.8.2
#> [79] reshape2_1.4.5 fgsea_1.36.2
#> [81] generics_0.1.4 gtable_0.3.6
#> [83] R.methodsS3_1.8.2 tidyr_1.3.2
#> [85] data.table_1.18.2.1 car_3.1-5
#> [87] XVector_0.50.0 BiocGenerics_0.56.0
#> [89] ggrepel_0.9.8 pillar_1.11.1
#> [91] stringr_1.6.0 limma_3.66.0
#> [93] yulab.utils_0.2.4 babelgene_22.9
#> [95] splines_4.5.3 tweenr_2.0.3
#> [97] treeio_1.34.0 lattice_0.22-9
#> [99] bit_4.6.0 tidyselect_1.2.1
#> [101] fontLiberation_0.1.0 GO.db_3.22.0
#> [103] locfit_1.5-9.12 Biostrings_2.78.0
#> [105] knitr_1.51 fontBitstreamVera_0.1.1
#> [107] bookdown_0.46 IRanges_2.44.0
#> [109] Seqinfo_1.0.0 edgeR_4.8.2
#> [111] SummarizedExperiment_1.40.0 stats4_4.5.3
#> [113] xfun_0.57 Biobase_2.70.0
#> [115] statmod_1.5.1 matrixStats_1.5.0
#> [117] stringi_1.8.7 lazyeval_0.2.2
#> [119] ggfun_0.2.0 yaml_2.3.12
#> [121] evaluate_1.0.5 codetools_0.2-20
#> [123] gdtools_0.5.0 qvalue_2.42.0
#> [125] BiocManager_1.30.27 ggplotify_0.1.3
#> [127] cli_3.6.5 systemfonts_1.3.2
#> [129] jquerylib_0.1.4 Rcpp_1.1.1
#> [131] png_0.1-9 parallel_4.5.3
#> [133] pkgdown_2.2.0 assertthat_0.2.1
#> [135] blob_1.3.0 clusterProfiler_4.18.4
#> [137] DOSE_4.4.0 tidytree_0.4.7
#> [139] ggiraph_0.9.6 scales_1.4.0
#> [141] purrr_1.2.1 crayon_1.5.3
#> [143] rlang_1.1.7 cowplot_1.2.0
#> [145] fastmatch_1.1-8 KEGGREST_1.50.0